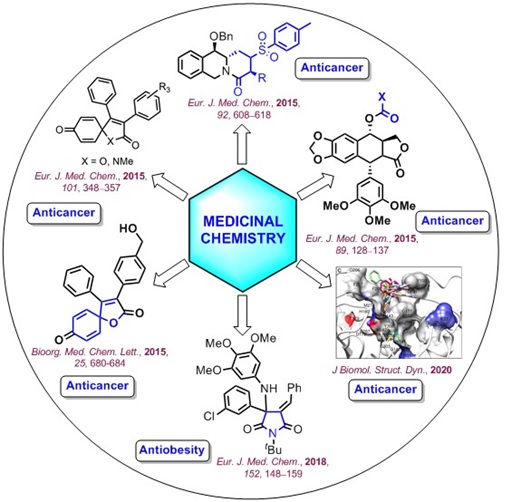
PDF) Design, synthesis, and structure-activity-relationship of phenyl imidazoles as potent Smoothened antagonists
![Synthesis of new chromeno-annulated cis-fused pyrano[4,3-c]isoxazole derivatives via intramolecular nitrone cycloaddition and their cytotoxicity evaluation – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on Synthesis of new chromeno-annulated cis-fused pyrano[4,3-c]isoxazole derivatives via intramolecular nitrone cycloaddition and their cytotoxicity evaluation – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on](https://cyberleninka.org/viewer_images/531223/f/1.png)
Synthesis of new chromeno-annulated cis-fused pyrano[4,3-c]isoxazole derivatives via intramolecular nitrone cycloaddition and their cytotoxicity evaluation – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on

LogD Contributions of Substituents Commonly Used in Medicinal Chemistry | ACS Medicinal Chemistry Letters

Bioorganic & Medicinal Chemistry Letters | Vol 14, Issue 9, Pages 2005-2374 (3 May 2004) | ScienceDirect.com by Elsevier
Writing Your Next Medicinal Chemistry Article: Journal Bibliometrics and Guiding Principles for Industrial Authors

Bioorganic & Medicinal Chemistry Letters | Recent Advances in Medicinal Chemistry | ScienceDirect.com by Elsevier
![PDF) Erratum to “Rational design of novel, potent piperazinone and imidazolidinone BACE1 inhibitors” [Bioorg. Med. Chem. Lett. 18 (2008) 3236–3241] | Jared Cumming and Andrew Stamford - Academia.edu PDF) Erratum to “Rational design of novel, potent piperazinone and imidazolidinone BACE1 inhibitors” [Bioorg. Med. Chem. Lett. 18 (2008) 3236–3241] | Jared Cumming and Andrew Stamford - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/41945149/mini_magick20190218-1404-1ork3dd.png?1550511543)
PDF) Erratum to “Rational design of novel, potent piperazinone and imidazolidinone BACE1 inhibitors” [Bioorg. Med. Chem. Lett. 18 (2008) 3236–3241] | Jared Cumming and Andrew Stamford - Academia.edu

Molecules | Free Full-Text | Synthesis and Biological Evaluation of Novel Benodanil-Heterocyclic Carboxamide Hybrids as a Potential Succinate Dehydrogenase Inhibitors

Bioorganic & Medicinal Chemistry Letters | PDF | Two Dimensional Nuclear Magnetic Resonance Spectroscopy | Nuclear Magnetic Resonance Spectroscopy
Bioorganic & Medicinal Chemistry Letters 1992: Vol 2 Index : Free Download, Borrow, and Streaming : Internet Archive
![PDF) Corrigendum to “Metal-assisted synthesis of unsymmetrical magnolol and honokiol analogs and their biological assessment as GABAA receptor ligands” [Bioorg. Med. Chem. Lett. 25/2 (2015) 400–403] PDF) Corrigendum to “Metal-assisted synthesis of unsymmetrical magnolol and honokiol analogs and their biological assessment as GABAA receptor ligands” [Bioorg. Med. Chem. Lett. 25/2 (2015) 400–403]](https://i1.rgstatic.net/publication/272568821_Corrigendum_to_Metal-assisted_synthesis_of_unsymmetrical_magnolol_and_honokiol_analogs_and_their_biological_assessment_as_GABAA_receptor_ligands_Bioorg_Med_Chem_Lett_252_2015_400-403/links/555b168208ae6fd2d8287b88/largepreview.png)
PDF) Corrigendum to “Metal-assisted synthesis of unsymmetrical magnolol and honokiol analogs and their biological assessment as GABAA receptor ligands” [Bioorg. Med. Chem. Lett. 25/2 (2015) 400–403]